Extruded fibers, loaded with precise dosages of drugs can be made into various implantable drug delivery platforms that can provide healing with more precision and efficacy than other methods now used. This is the premise of Dallas, Texas-based TissueGen Inc., the developer of the absorbable polymer technology.
In a presentation at IFAI’s Advanced Textiles Expo in October, the company’s CSO Dr. Kevin Nelson explained the breakthrough technology and its commercial viability.
According to Nelson, the patented process enables fiber extrusion at room temperature with drugs and therapeutic agents previously impossible to incorporate in implant devices. Additional advantages of drug delivery from fiber are that the fibers are mechanically strong, remain in place, and offer uniform diameter and drug concentration, and slower release than spheres of the same diameter. Further, the devices can be readily mass produced.
Overcoming the challenges
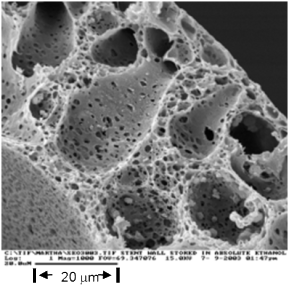
Of the fiber manufacturing methods (melt extrusion, electrospinning and wet extrusion), the latter offers the strongest fibers and the widest range of drugs that can be used. However, this method uses harsh organic solvents. Very few drugs can survive extrusion and the residual solvents must be removed from the fiber.
To address this, the company developed a process that creates a protective “bubble” for the drug in the solvent bath, the drug-loaded polymer is then extruded and a post process removes residual solvents. Pharmaceuticals and biologically-derived agents can be loaded using this process. This includes antibiotics, antimicrobials and cancer remediation pharmaceuticals, as well as a variety of proteins.
Using a variable pitched coil delivery format, the process can direct cells to migrate through specified pathways and provide for a “tunable,” controlled drug delivery.
Release studies
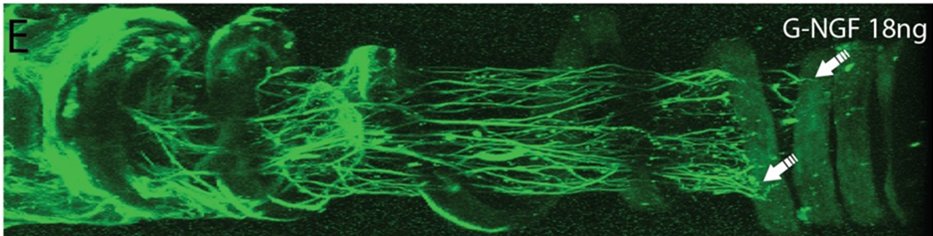
For example, in a In Vivo animal study, a monofilament fiber was loaded with Aldose Reductase Inhibitor (ARI), which blocks conversion of glucose to sorbitol, a potential cause of blindness in diabetic patients. A single dose over six months resulted in a five-fold reduction of diabetic state indicators in diabetic rats.
The site-specific, small-dose drug delivery is the only way this can be accomplished, because systemic delivery would likely be fatal. Other studies have had success in solid pancreatic tumor remediation in rats.
Now and in the future
The company expects this drug delivery material will assist with nerve regeneration, tissue engineering, and in medical textiles for hernia mesh, pouches and slings, and tendon and ligament repair.
A biodegradable drug-loaded (BDL) fiber trademarked ELUTE® is commercially available for pharmaceuticals and BDAs.
 TEXTILES.ORG
TEXTILES.ORG


